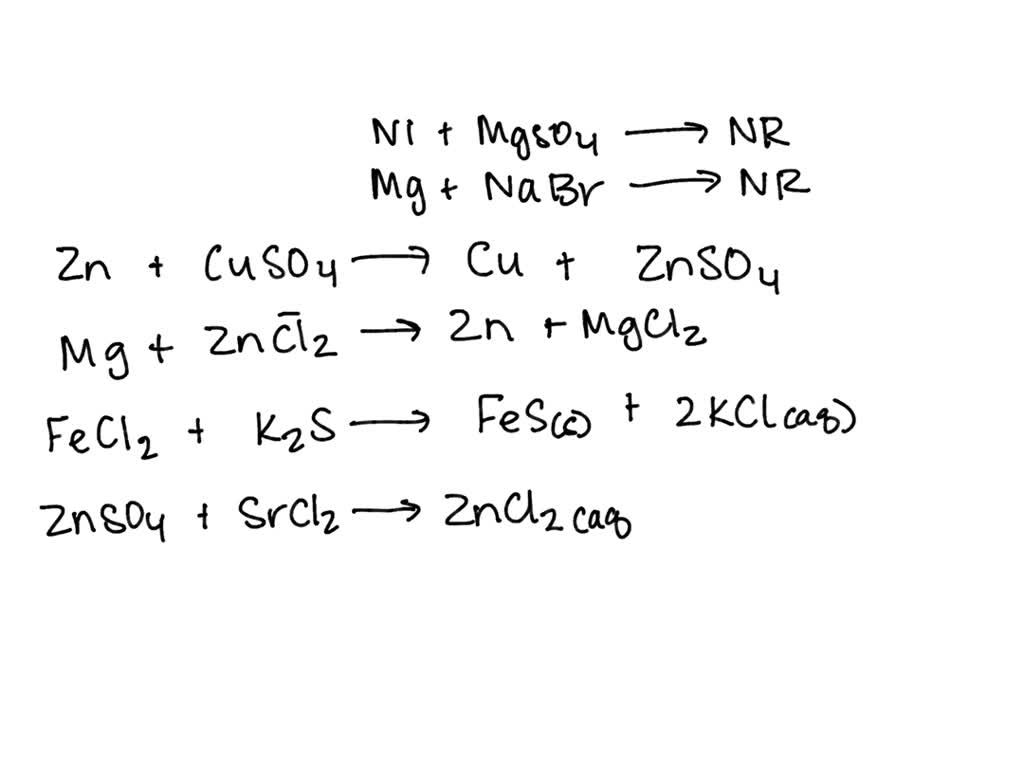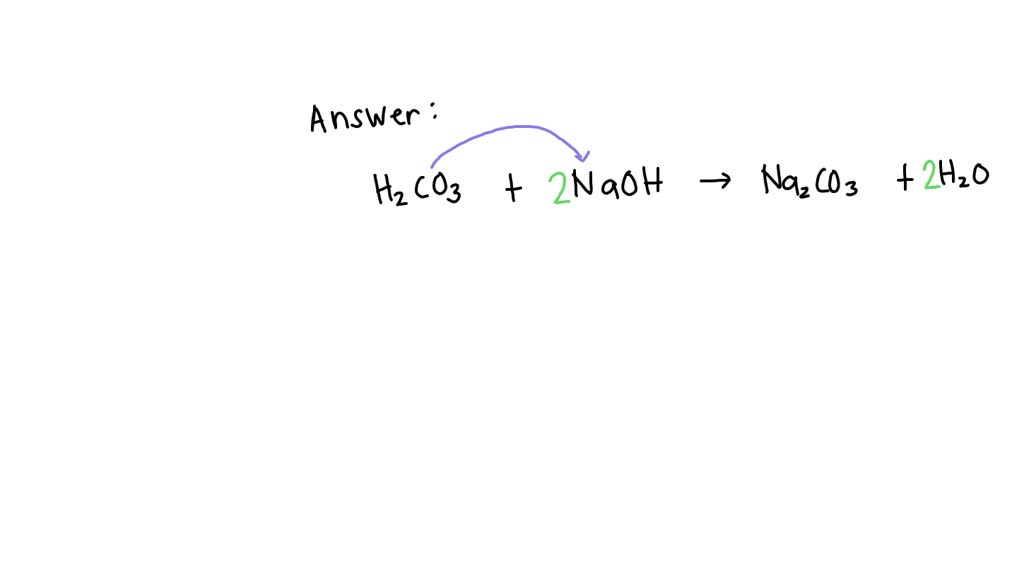What Type Of Reaction Is Na2Co3 Hcl
What Type Of Reaction Is Na2Co3 Hcl - Na 2 co 3 + hcl → nacl + co 2 + h 2 o. This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial. 2hcl +nax2cox3 2nacl +hx2o +cox2 2 h c l + n a x 2 c o x 3 2 n a c l + h x 2 o + c o x 2. In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. Na2co3 (s) + hcl (aq) = nacl (aq) + h2o (l) + co2 (g) the word equation for the reaction is: Balance the reaction of na2co3 + hcl = nacl + h2o + co2 using this chemical equation balancer!
2hcl +nax2cox3 2nacl +hx2o +cox2 2 h c l + n a x 2 c o x 3 2 n a c l + h x 2 o + c o x 2. Na2co3 (s) + hcl (aq) = nacl (aq) + h2o (l) + co2 (g) the word equation for the reaction is: We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial. Balance the reaction of na2co3 + hcl = nacl + h2o + co2 using this chemical equation balancer! In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. Na 2 co 3 + hcl → nacl + co 2 + h 2 o.
Balance the reaction of na2co3 + hcl = nacl + h2o + co2 using this chemical equation balancer! We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. Na2co3 (s) + hcl (aq) = nacl (aq) + h2o (l) + co2 (g) the word equation for the reaction is: This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. 2hcl +nax2cox3 2nacl +hx2o +cox2 2 h c l + n a x 2 c o x 3 2 n a c l + h x 2 o + c o x 2.
Sodium Carbonate to Produce Sodium Oxide and Carbon Dioxide
Balance the reaction of na2co3 + hcl = nacl + h2o + co2 using this chemical equation balancer! We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. This chemistry video tutorial explains how to predict the products of.
SOLVED Please Complete the balance and label the raction type Complete
This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. 2hcl +nax2cox3 2nacl +hx2o +cox2 2 h c l + n a x 2 c o x 3 2 n a c l + h x 2 o + c o x 2. Na2co3 (s) + hcl (aq) = nacl (aq).
Balanced Equation of Sodium Carbonate and Hydrochloric Acid
Na 2 co 3 + hcl → nacl + co 2 + h 2 o. 2hcl +nax2cox3 2nacl +hx2o +cox2 2 h c l + n a x 2 c o x 3 2 n a c l + h x 2 o + c o x 2. We will discuss about different characteristics of sodium carbonate and hcl acid.
SOLVED H2CO3 + NaOH → Na2CO3 + H2O identify its type of reaction
2hcl +nax2cox3 2nacl +hx2o +cox2 2 h c l + n a x 2 c o x 3 2 n a c l + h x 2 o + c o x 2. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. Na2co3 (s) + hcl (aq) = nacl (aq) + h2o (l) +.
`Na_(2)CO_(3)+HCl to ………. + …….. + ………` The products in the above
In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. We.
Na2CO3+HCl=NaCl+CO2+H2O Balanced EquationSodium carbonate
In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. We.
CHEMICAL REACTION Na2CO3 + 2HCl Unleashed
Na 2 co 3 + hcl → nacl + co 2 + h 2 o. Na2co3 (s) + hcl (aq) = nacl (aq) + h2o (l) + co2 (g) the word equation for the reaction is: We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial. This chemistry video tutorial explains how to predict.
SOLVED Indicates the type of reaction Na2CO3 + HCl → NaCl + H2CO3 show
Na 2 co 3 + hcl → nacl + co 2 + h 2 o. Balance the reaction of na2co3 + hcl = nacl + h2o + co2 using this chemical equation balancer! In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. This chemistry.
SOLVED Reaction Na2CO3(aq) + CaCl2(aq) → CaCO3(s) + 2NaCl(aq) Mass
We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial. In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. Na2co3 (s) + hcl.
PPT Writing Chemical Reactions PowerPoint Presentation, free download
This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. Na 2 co 3 + hcl → nacl + co 2 + h 2 o. Balance.
Na 2 Co 3 + Hcl → Nacl + Co 2 + H 2 O.
In this video we determine the type of chemical reaction for the equation na2co3 + hcl = nacl + h2o + co2 (sodium carbonate +. This chemistry video tutorial explains how to predict the products of the reaction between sodium carbonate and hydrochloric acid. Na2co3 (s) + hcl (aq) = nacl (aq) + h2o (l) + co2 (g) the word equation for the reaction is: Balance the reaction of na2co3 + hcl = nacl + h2o + co2 using this chemical equation balancer!
2Hcl +Nax2Cox3 2Nacl +Hx2O +Cox2 2 H C L + N A X 2 C O X 3 2 N A C L + H X 2 O + C O X 2.
We will discuss about different characteristics of sodium carbonate and hcl acid reaction in this tutorial.