Gamp 5 Full Form
Gamp 5 Full Form - Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. What are gamp® 5 categories, requirements, and. The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems. Good automated manufacturing practice, founded in 1991. Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the.
Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. Good automated manufacturing practice, founded in 1991. What are gamp® 5 categories, requirements, and. The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for.
What are gamp® 5 categories, requirements, and. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. Good automated manufacturing practice, founded in 1991. Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems.
GAMP 5 and GAMP 5 2nd Edition What are the main differences? Scilife
Good automated manufacturing practice, founded in 1991. What are gamp® 5 categories, requirements, and. The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Gamp 5, which stands for good automated manufacturing practice, is a set of.
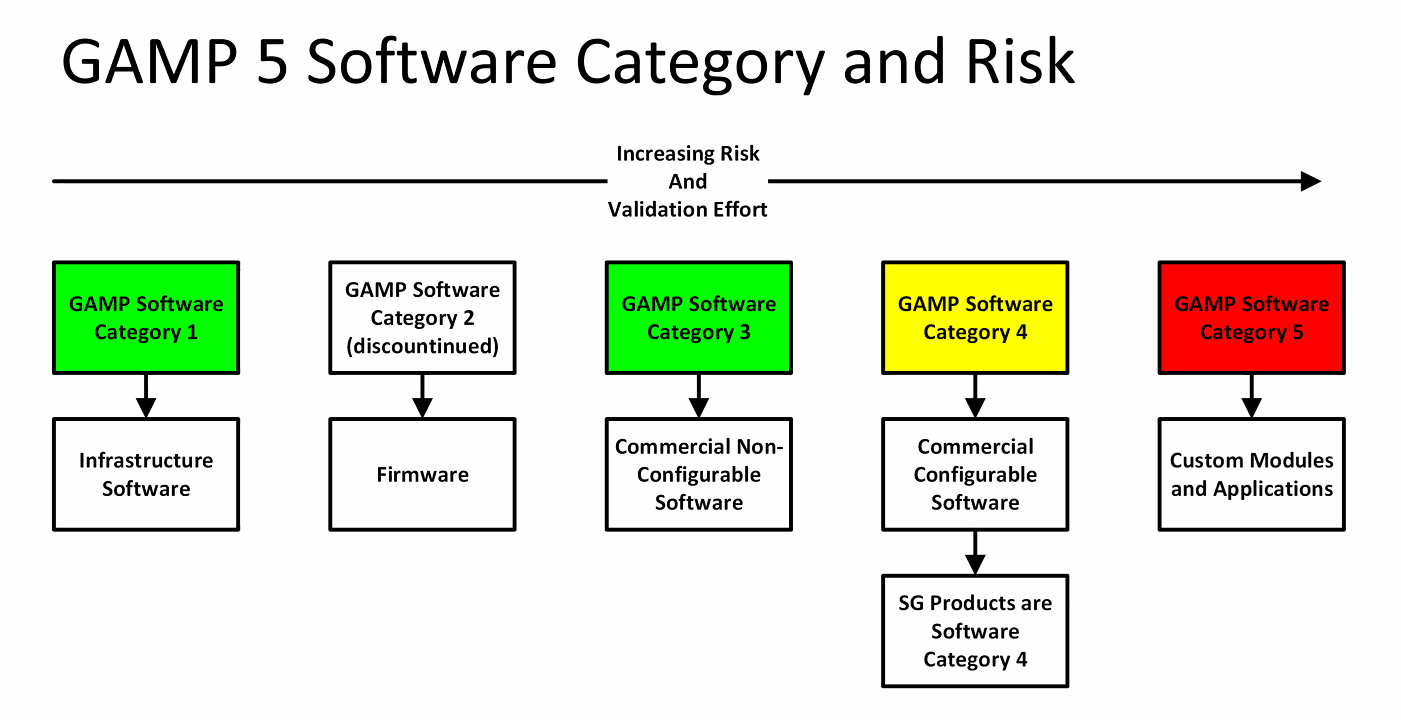
V5 GAMP 5 Software Category & Risk SG Systems Global
Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. Good automated manufacturing practice, founded in 1991. What are gamp® 5 categories, requirements, and. Gamp 5 provides a structured.
GAMP 5 Uncovered What You Need to Know Scilife
The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems. Good automated manufacturing practice, founded in 1991. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. What are gamp® 5 categories, requirements, and. Gamp (good automated manufacturing practice) is a set of guidelines and best practices.
GAMP 5 and GAMP 5 2nd Edition What are the main differences? Scilife
Good automated manufacturing practice, founded in 1991. What are gamp® 5 categories, requirements, and. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. Gamp 5, which stands for.
GAMP5 GUIDELINES PDF
Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems. Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. Good automated manufacturing practice, founded in.
GAMP 5 and GAMP 5 2nd Edition What are the main differences? Scilife
The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems. Good automated manufacturing practice, founded in 1991. What are gamp® 5 categories, requirements, and. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. Gamp 5 provides a structured approach to the validation of computerized systems used.
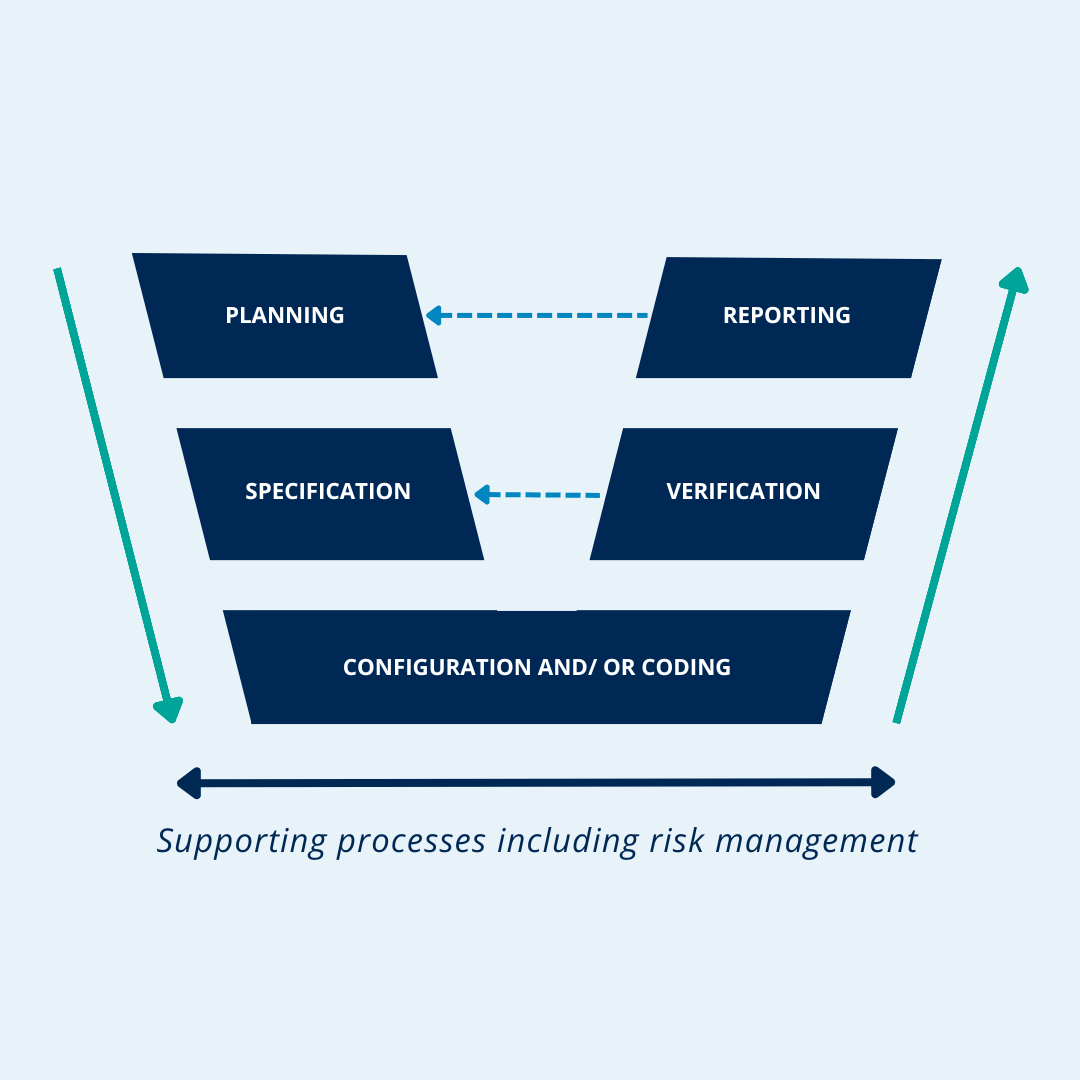
What is the GAMP 5 Vmodel in CSV? QbD Group
Good automated manufacturing practice, founded in 1991. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. Gamp 5, which stands for good automated manufacturing practice, is a set.
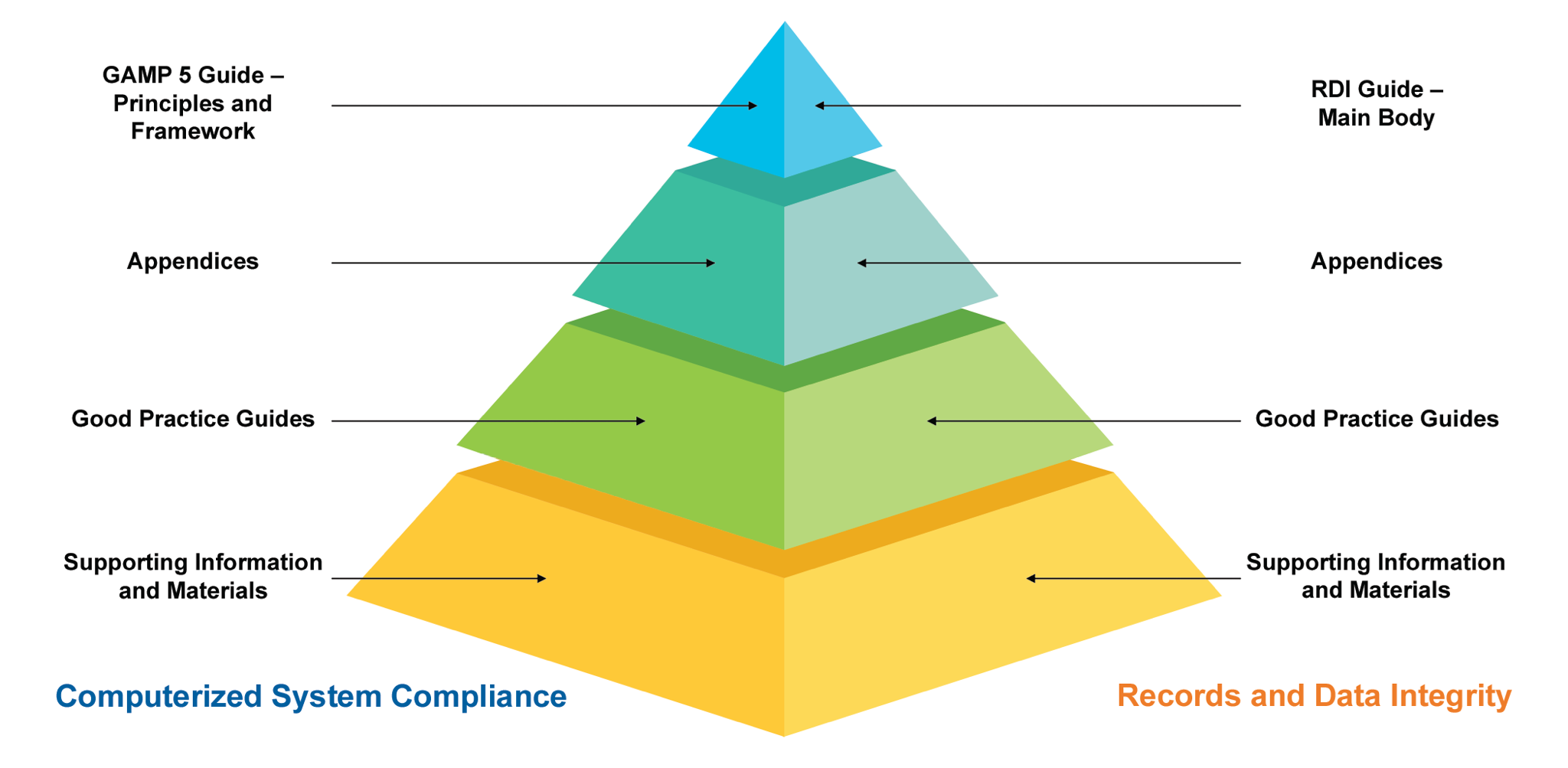
ISPE GAMP5 second edition What’s new?
What are gamp® 5 categories, requirements, and. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. Good automated manufacturing practice, founded in 1991. Gamp (good automated manufacturing practice) is.
GAMP 5 Uncovered What You Need to Know Scilife
What are gamp® 5 categories, requirements, and. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. Good automated manufacturing practice, founded in 1991. Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. Gamp 5 provides a structured.
What is the GAMP 5 Vmodel in CSV? QbD Group
Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. What are gamp® 5 categories, requirements, and. Good automated manufacturing practice, founded in 1991. Gamp 5 provides a structured approach to the validation of computerized systems used in the manufacturing and testing of pharmaceutical. The gamp® 5 guidelines are essential.
Gamp 5 Provides A Structured Approach To The Validation Of Computerized Systems Used In The Manufacturing And Testing Of Pharmaceutical.
Gamp (good automated manufacturing practice) is a set of guidelines and best practices for the validation of automated systems in the. What are gamp® 5 categories, requirements, and. Gamp 5, which stands for good automated manufacturing practice, is a set of guidelines created by the international society for. The gamp® 5 guidelines are essential for ensuring compliant, efficient pharma manufacturing systems.