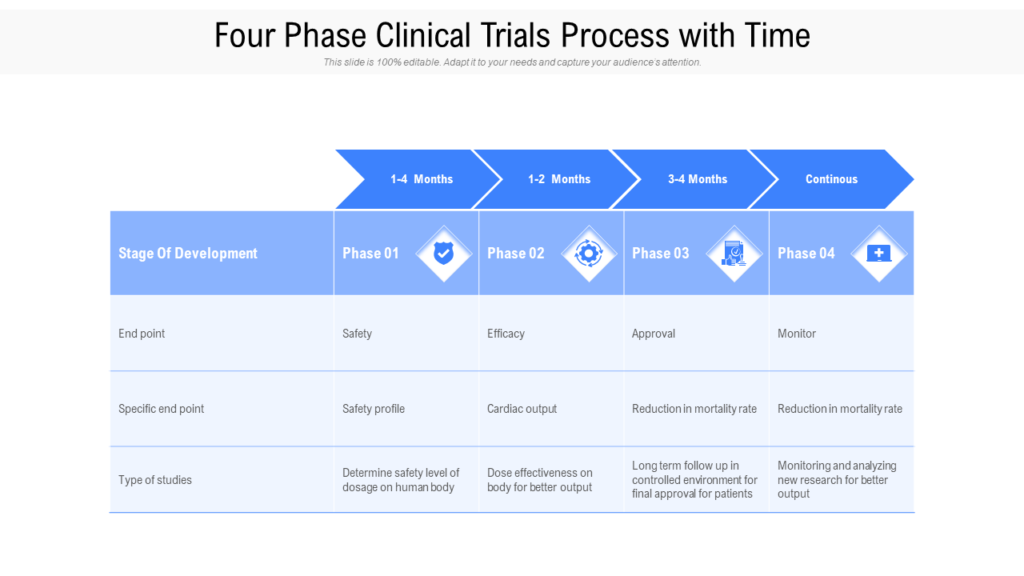
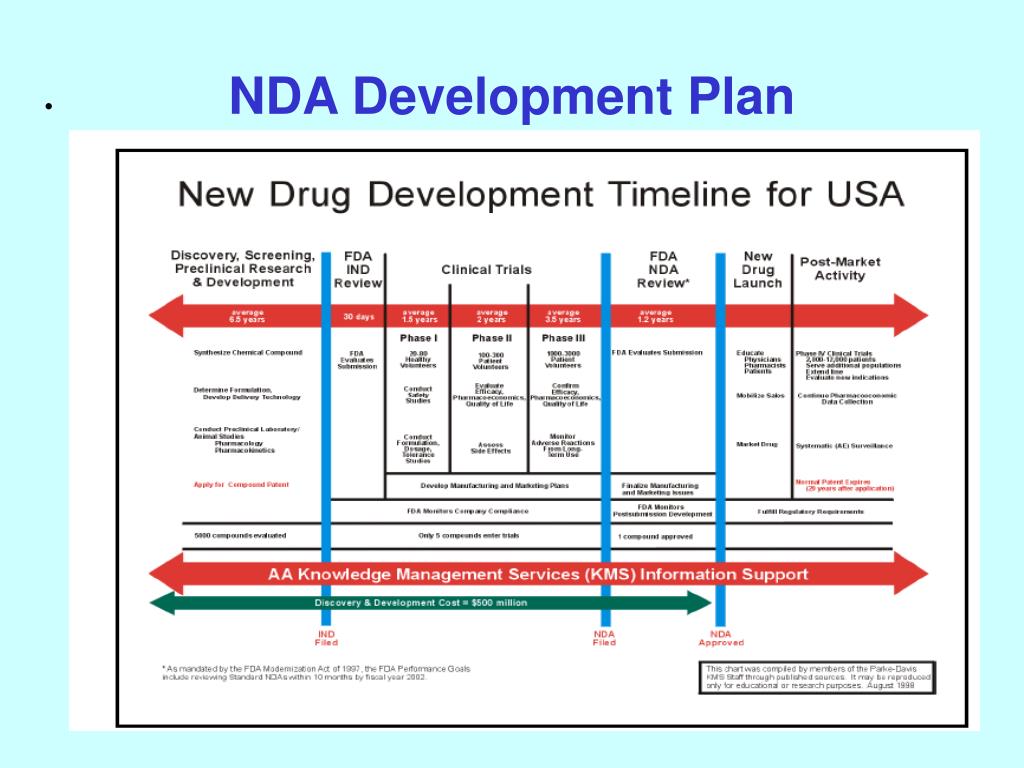
Clinical Development Plan Template Fda
Clinical Development Plan Template Fda - 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator This template is to help behavioral and social science researchers. Guidance for industry content and format of investigational new drug applications (inds). A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Ind content and format for phase 1 studies. Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism
Ind content and format for phase 1 studies. Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. This template is to help behavioral and social science researchers. A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism Guidance for industry content and format of investigational new drug applications (inds). 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator
Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator Guidance for industry content and format of investigational new drug applications (inds). Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism This template is to help behavioral and social science researchers. A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Ind content and format for phase 1 studies.
Clinical Development Plan Template
This template is to help behavioral and social science researchers. Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Ind content and format for phase 1 studies. Guidance for industry.
30 Clinical Development Plan Template Hamiltonplastering
Ind content and format for phase 1 studies. Guidance for industry content and format of investigational new drug applications (inds). This template is to help behavioral and social science researchers. 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process.
Early Clinical Development Plan Introduction EUPATI Open Classroom
This template is to help behavioral and social science researchers. A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Guidance for industry content and format of investigational new drug applications (inds). Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and.
Clinical Development Plan Template
Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary.
OnePager Clinical Development Plan
1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and.
Clinical Development Plan Template
A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Ind content and format for phase 1 studies. This template is to help behavioral and social science researchers. Guidance for industry content and format of investigational new drug applications (inds). 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator
Clinical Development Plan Template Fda
Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. This template is to help behavioral and social science researchers. 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism Guidance for industry content and format of investigational.
Clinical Development Plan Template Venngage
1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator Ind content and format for phase 1 studies. This template is to help behavioral and social science researchers. A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Download the available cdp template to facilitate the multidisciplinary development of a clinical.
Clinical Development Plan Template
1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator This template is to help behavioral and social science researchers. Ind content and format for phase 1 studies. Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism
Clinical Development Plan Template
Clinical investigational plan synopsis • new onset atrial fibrillation • pulmonary embolism 1571, 1572, 3454, 3455, financial, disclosure, certification, form, clinical, investigator A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and.
Clinical Investigational Plan Synopsis • New Onset Atrial Fibrillation • Pulmonary Embolism
Download the available cdp template to facilitate the multidisciplinary development of a clinical program that is robust, feasible and aligned. Ind content and format for phase 1 studies. A target product profile (tpp) is a planning tool introduced by the fda to streamline the drug development process. Guidance for industry content and format of investigational new drug applications (inds).
1571, 1572, 3454, 3455, Financial, Disclosure, Certification, Form, Clinical, Investigator
This template is to help behavioral and social science researchers.
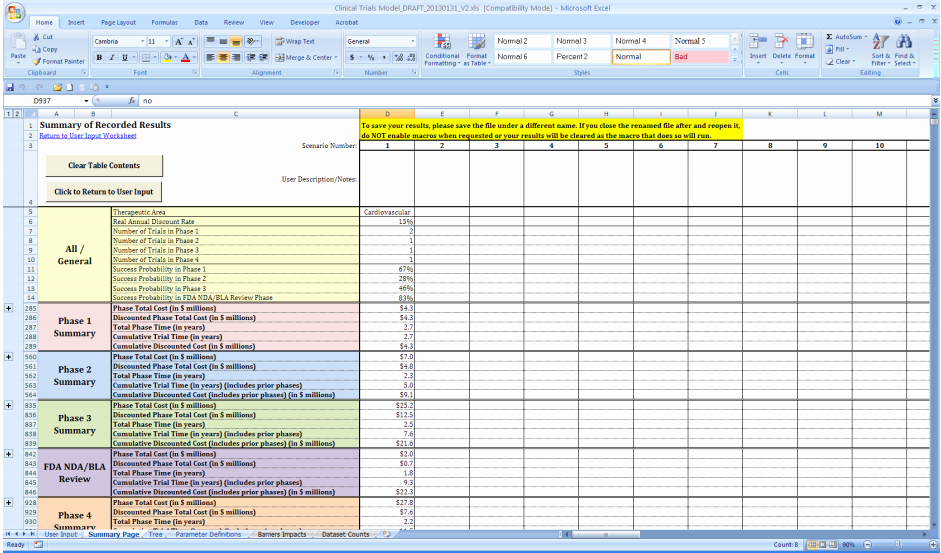
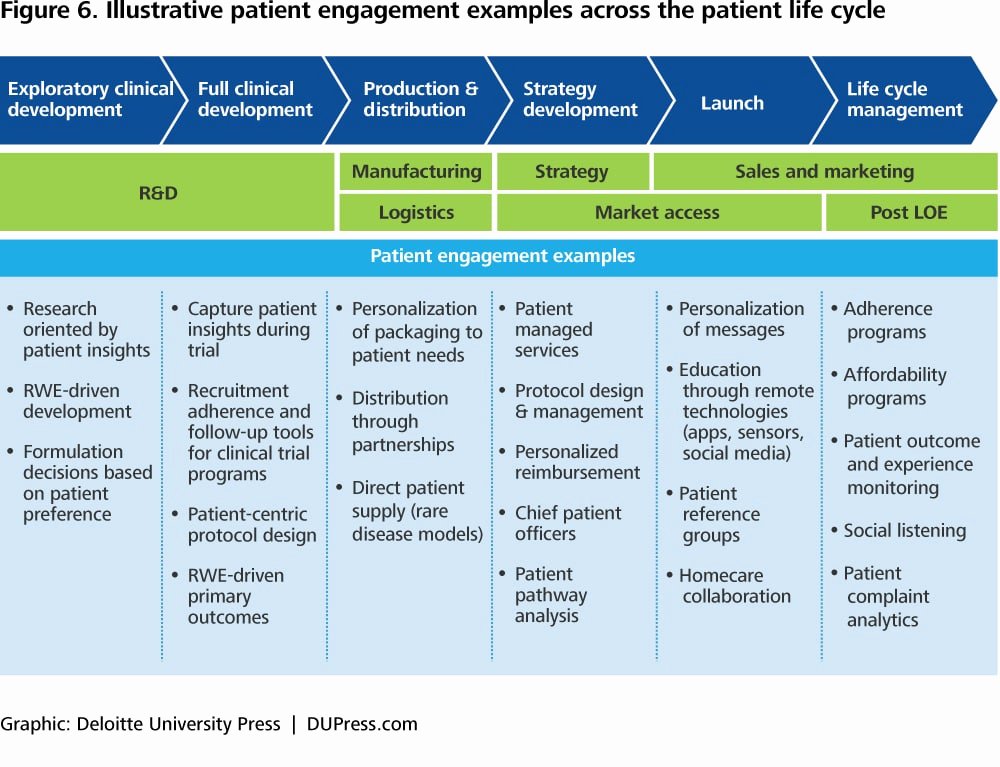
.jpg)